Swimming pool chlorine refers to the chemical compound used to sanitize and disinfect pool water. It is commonly used in the swimming pool and spa industry as the primary sanitizer. Chlorine emerges in different forms, such as gas chlorine (chlorine gas), liquid chlorine (bleach or sodium hypochlorite), or chlorinating granules (such as sodium dichloroisocyanurate). Chlorine helps control the growth of bacteria and other organisms in the pool water.
Chlorine is a greenish-yellow gas that is 2.5 times heavier than air. Chlorine belongs to the Group-VIIA elements on the Periodic Table, also known as halogens. Chlorine is not found in its pure form in nature and makes up 0.15 percent of the earth’s crust, primarily as soluble chlorides like sodium chloride (NaCl). Chlorine is a common disinfectant used in swimming pools and can inactivate or kill a vast majority of disease-causing microorganisms. Chlorine is grouped into three categories: elemental, inorganic, and organic.
Chlorine is a gas with a CAS number of 7782505 and a UN ID number of UN2465. Its chemical formula is Cl. It does not contain any stabilizer and does not increase cyanuric acid per pound in 10,000 gallons.
Contents
- What is Disinfectant (Sanitizer)?
- What are the types of chlorine?
- What is Gas Chlorine?
- What is Sodium Hypochlorite?
- What is Calcium Hypochlorite?
- What is Lithium Hypochlorite?
- What is Trichlor?
- What is Dichlor?
- What are the alternatives to Chlorine for pools?
- What is Oxidation?
- What is chlorine generation?
- What is Chlorine Dioxide?
- What is active chlorine?
- What is available chlorine content?
- What is free chlorine?
- What is Combined Chlorine or Chloramines?
- What is active chlorine percentage?
- How does chlorine interact with other chemicals in the water?
- What are the Properties of Chlorine?
- What are the costs of different forms of chlorine?
- What is Breakpoint Chlorination?
- What does chlorine produce when added to pool water?
- What are the Evaluation Criteria for Chlorine?
- What is an ideal Chlorine level for swimming pools?
- What are the signs of low Chlorine in the pool?
- What are the consequences of swimming in a pool with low Chlorine?
- How to increase Chlorine in the pool?
- What are the effects of low Chlorine in pool water?
- How to lower pool Chlorine?
- What are the effects of Chlorine on pool water quality?
- What is Chlorine in chemistry?
- What is the importance of Chlorine for pool water?
- How to handle Chlorine safely when treating your pool?
- What is the Effect of Chlorine Combining with CYA?
- Are the terms hypochlorous acid and chlorine used interchangeably?
- Do disinfectants release chlorine in the pool?
- What are the categories of chlorinating agents?
- What happens to chlorine when it is used up?
- How does Chlorine interact with CYA?
- How are Chlorine Products Labeled?
- What are supplemental disinfectants?
What is Disinfectant (Sanitizer)?
A Disinfectant, also known as a Sanitizer, is an agent that destroys disease-carrying microorganisms. Disinfectants are substances that inactivate or kill over 99.9% of disease-causing microorganisms, including bacteria, fungi, viruses, and protozoan parasites. Disinfectans and Sanitizers are also effective against algae. The term “inactivate” is used because some pathogens, like viruses or microscopic cysts, are not alive and therefore cannot be killed. However, they can be rendered non-infectious.
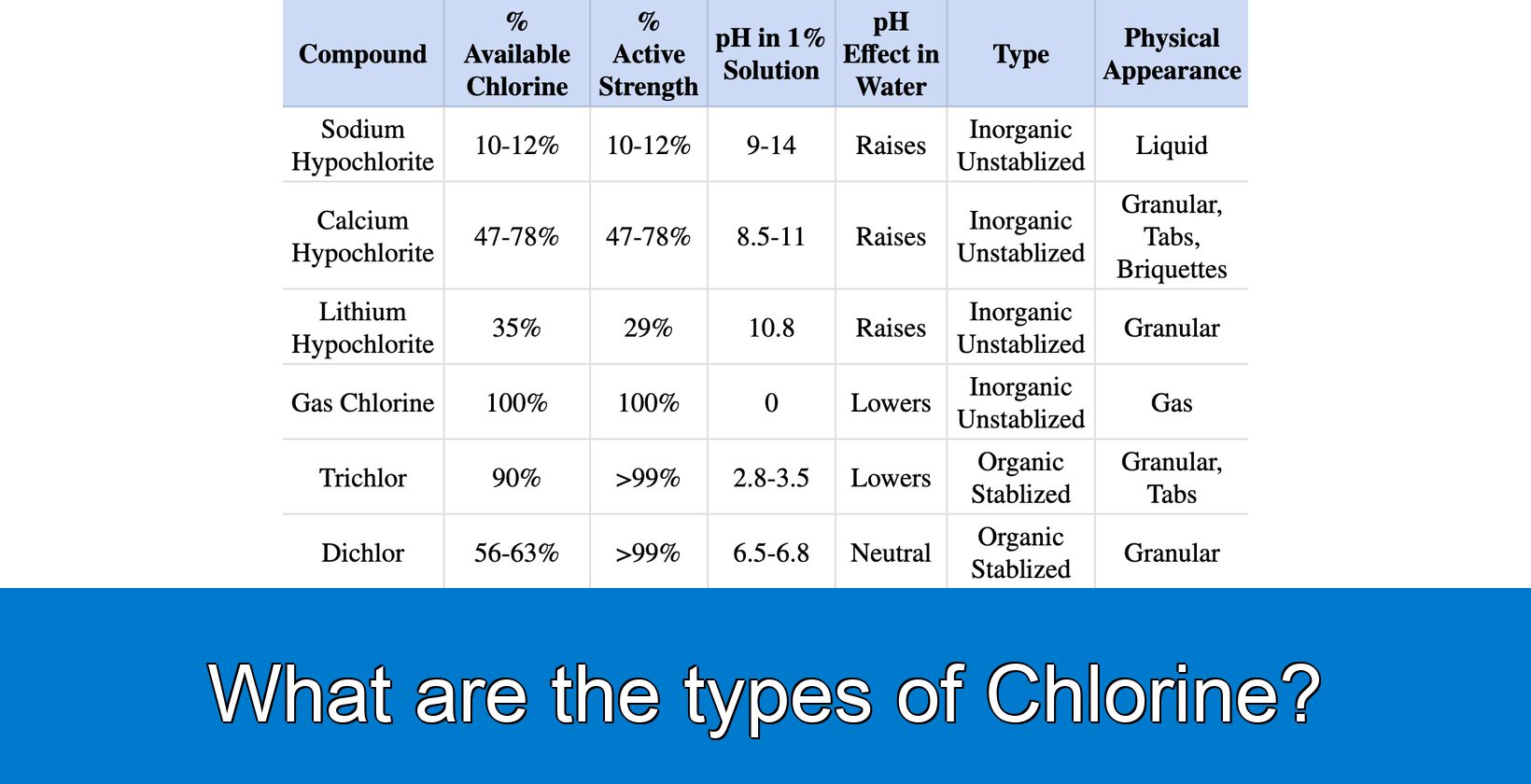
What are the types of chlorine?
The types of chlorine are elemental, inorganic, and organic. Elemental chlorine is Chlorine, element number 17 on the Periodic Table. Chlorine is a gas under standard conditions, often referred to as gas chlorine in the pool industry. Inorganic chlorine includes sodium hypochlorite (liquid chlorine), calcium hypochlorite (cal hypo), and lithium hypochlorite (lithium hypo). Organic chlorine refers to chlorine compounds with man-made chemicals, also known as stabilized chlorine, chlorinated isocyanurates, or chlorinated isos. Specifically, chlorine compounds are sodium dichloroisocyanurate and trichloroisocyanurate (dichlor and trichlor).
What is Gas Chlorine?
Gas chlorine is an unstabilized, inorganic compound, that exists in a gaseous state. This greenish-yellow colored compound is 2.5 times heavier than air. Gas chlorine possesses 100% available chlorine and the same percentage of active strength as liquid chlorine. When marketers use this compound in a 1% solution, the pH lowers to 0. Despite the hazardous nature of gas chlorine, which causes irritation to the eyes, nose, and throat, and is lethal in extreme cases, marketers find this compound popular due to its cost-effectiveness.
Gas chlorine dissolves highly in water, making this compound an effective disinfectant for pool water cleaning. Electrolytic cells primarily produce gas chlorine. In the context of pool water cleaning, gas chlorine performs the same primary functions as other forms of chlorine, namely disinfection and oxidation. A gas chlorinator directly injects gas chlorine into the pool water. This method is employed in larger commercial or public pools due to the compound’s ability to treat large volumes of water quickly.
However, because of the hazardous nature of gas chlorine, this compound necessitates careful handling and storage. Trained professionals often manage gas chlorine to ensure safety. Regardless of the form of chlorine used, maintaining proper chlorine levels in the pool water is crucial. The recommended chlorine level for swimming pools typically ranges from 1.0 to 3.0 parts per million (ppm).
What is Sodium Hypochlorite?
Sodium hypochlorite (NaOCl) is an unstabilized, inorganic compound in liquid form, having a chlorine availability and active strength of 10-12%. This compound serves as a vital source of chlorine in the context of pool cleaning, performing several key roles. Sodium Hypochlorite disinfects by releasing free chlorine into the pool water. Sodium Hypochlorite effectively kills harmful microorganisms and ensures the safety of the water for swimming.
Moreover, Sodium Hypochlorite acts as an oxidizer in the pool water, breaking down organic matter and other contaminants, which facilitates their easier removal. The chlorine that Sodium Hypochlorite releases also aids in preventing the growth of algae. Algae cloud the water and make pool surfaces slippery. Sodium hypochlorite, with its high pH level, also balances acidic pool water.
However, maintaining a consistent monitoring and adjustment of the pool’s pH level is crucial to preserve the effectiveness of chlorine as a disinfectant. In summary, Sodium hypochlorite is a potent agent in pool cleaning due to its high chlorine availability and active strength. Sodium hypochlorite disinfects, oxidizes, prevents algae, and regulates pH level.
What is Calcium Hypochlorite?
Calcium Hypochlorite, also known as Cal Hypo, is an unstabilized, inorganic compound that acts as a chlorine source in the cleaning of swimming pools. When introduced to pool water, Calcium Hypochlorite reacts to form hypochlorous acid and hypochlorite ions, which are collectively referred to as free chlorine. Free chlorine disinfects the pool by killing bacteria and viruses. Free chlorine also oxidizes organic and inorganic matter. Organic and inorganic matter make the water cloudy and cause the growth of unwanted microorganisms.
Moreover, Calcium Hypochlorite adds to the residual chlorine in the water, which continues to sanitize the pool and provide protection against contamination. Pool cleaners use Calcium Hypochlorite for shock treatment or superchlorination of pool water, because of the high available chlorine content and moderate solubility of Cal Hypo. This treatment method raises the chlorine level, kills all remaining microorganisms, and breaks down chloramines that cause eye irritation and a strong chlorine odor.
In conclusion, Cal Hypo is essential in pool cleaning, because it provides a high available chlorine content for disinfection, oxidation, and shock treatment. Due to Calcium Hypochlorite the swimming environment remains clean and safe.
What is Lithium Hypochlorite?
Lithium Hypochlorite, also known as Lithium Hypo, is an unstabilized, inorganic compound that appears as a granular substance or a white powder. Lithium Hypochlorite boosts the pool water with 35% available chlorine and has a potent composition with an active strength of 29%. Lithium Hypo is highly alkaline. When a solution contains 1% Lithium Hypochlorite, its pH level rises to 10.8. Unlike other pool sanitizing agents, Lithium Hypo does not contain stabilizers such as cyanuric acid or conditioner.
The manufacturing process of Lithium Hypochlorite involves the mixing of chlorine gas with caustic soda. This mixture then reacts with lithium hydroxide, resulting in a product with high available chlorine content, which is crucial for effective pool disinfection. A key advantage of Lithium Hypochlorite is its high solubility. Lithium Hypo dissolves quickly when distributed over the surface of the pool. This rapid dissolution makes Lithium Hypochlorite a highly suitable option for pools that have vinyl lining or dark plaster.
Lithium Hypochlorite dissolves quickly, leading to a fast boost in the pool’s chlorine level. This makes it an appropriate choice for shock treatments in swimming pools.
What is Trichlor?
Trichlor, known as trichloro-s-triazinetrione or trichloroisocyanuric acid, is a stabilized, organic chlorine compound in the process of pool cleaning. This compound is available in granular form or tabs. Trichlor introduces 90% of available chlorine into the pool water, more than any other chlorine compound. This means that Trichlor is a highly effective sanitizer, because it possesses more than 99% active strength.
Trichlor is a white, slow-dissolving compound that has an acidic pH, which ranges between 2.8 and 3.0. When applied in a 1% solution, Trichlor reduces the pool water’s pH to a range of 2.8 to 3.5. This helps keep the water clear and prevents it from becoming cloudy. An important attribute of Trichlor is its inherent stabilizer. This feature protects the chlorine from degradation by sunlight, thereby extending its effectiveness.
Trichlor’s high chlorine content and acidic properties make Trichlor suitable for regular chlorination, but it’s not the optimal choice for superchlorination. Consequently, regular monitoring and adjustment of the pool’s pH and chlorine levels are necessary when using Trichlor, to ensure the water remains safe and comfortable for swimming.
What is Dichlor?
Dichlor is an organic compound that serves as a stable source of chlorine for sanitizing pools. Dichlor offers a chlorine availability range of 56-63% and maintains an active ingredient concentration of over 99%. When Dichlor is added to pool water, Dichlor releases chlorine that acts as a disinfectant, wiping out bacteria, viruses, and other microorganisms. Dichlor also includes cyanuric acid, a component that protects the released chlorine from sunlight degradation. For this reason, Dichlor is suitable for outdoor pools. When a solution contains 1% Dichlor, the pH ranges between 6.5 and 6.8. The neutral pH stops the pool water from turning too acidic or alkaline. Dichlor is also used for shock treatments, which temporarily boost chlorine levels to destroy bacteria and combined chlorine compounds.
What are the alternatives to Chlorine for pools?
The 5 alternatives to chlorine for pools include bromine, ozone, UV, AOP (Advanced Oxidation Process) and Biguanicides.
- Bromine is an alternative sanitizer and disinfectant. Bromine is effective in killing bacteria and viruses and is more stable than chlorine in higher temperatures. Bromine is used in hot tubs and spas. Bromine (BCDMH) is a stabilized, organic compound that appears in granular form or tabs. It has 27% available chlorine and 95.5% active strength. In a 1% solution, Bromine lowers the pH to 4.8.
- Ozone is another alternative that is used to improve water quality and reduce the amount of chlorine needed. Ozone replaces 50 to 90 percent of chlorine or bromine in a pool or spa, resulting in superior water quality.
- UV (Ultraviolet) treatment is a method that uses UV light to kill bacteria, viruses, and other microorganisms in the water. UV (Ultraviolet) is used in conjunction with a small amount of chlorine or bromine to maintain residual disinfection.
- AOP (Advanced Oxidation Process) is a combination of ozone and UV treatment. It produces hydroxyl radicals that effectively oxidize and destroy contaminants in the water. AOP (Advanced Oxidation Process) reduces chlorine consumption by up to 90% if sized correctly.
- Biguanicides, known by the trade name Baquacil, are a type of disinfectant polymer. Biguanicides are effective sanitizers but not oxidizers, so they can’t be used alone. Biguanicides require a hydrogen peroxide product for monthly shock and a quaternary ammonium-based supplement weekly. Biguanicides can’t be mixed with chlorine products or any other chemicals except those designed as part of the package.
These are options for pool owners who prefer alternatives to chlorine or want to reduce their chlorine usage. The selection of a particular method depends on various factors such as budget, water properties, and the number of swimmers.
What is Oxidation?
Oxidation is a chemical process used in swimming pools to remove organic waste and kill bacteria. Chlorine, a common disinfectant used in swimming pools, is involved in this process. Oxidation effectively kills pathogens and algae, and also oxidizes other materials. Approximately 85% of the chlorine added to pool water is used for oxidation, not for killing organisms. Chlorine has an oxidizing power, which is determined by analyzing the amount of free chlorine.
What is chlorine generation?
Chlorine generation is the process of producing chlorine gas, most often through electrolysis. Chlorine generation involves using a chlorine generator that uses salt, electricity, and water.
What is Chlorine Dioxide?
Chlorine dioxide is a selective oxidizing agent and disinfectant used to kill bacteria, algae, viruses, and microscopic cysts like Cryptosporidium or Giardia.
What is active chlorine?
Active chlorine refers to the weight percentage of the active ingredient in a product. It is the active disinfectant in the water, often in the form of hypochlorous acid (HOCl).
What is available chlorine content?
Available chlorine content (ACC) refers to the relative amount of chlorine released into water by different chlorine disinfectants, using chlorine gas as the standard reference of 100%. It is a term used to compare the disinfecting power of different chlorine compounds. The oxidizing power is determined by analyzing the amount of chlorine available for oxidizing.
What is the Difference Between Available and Active Chlorine?
Understanding the difference between available chlorine content (ACC) and active chlorine percentage is crucial. ACC refers to the relative amount of chlorine released into water by different chlorine disinfectants, using chlorine gas as the standard reference of 100%. On the other hand, active chlorine percentage refers to the weight percentage of the active ingredient in the product. For instance, a 100-pound container of trichlor would contain roughly 100 pounds of actual trichlor, as the active ingredient percentage is typically over 99%. When considering ACC, pure trichlor has an ACC of 90%.
What is free chlorine?
Free chlorine (FC) is the active disinfectant in the water. It is the sum of HOCI and OCI and is determined by the DPD test. The common practice in pools is to maintain the free chlorine at 2.0 to 4.0 ppm. The accepted minimum is 1.0 ppm in the U.S., and the maximum is subject to debate.
What is the Required Level of Free Chlorine?
The required level of free chlorine according to the APSP and most guidelines is a minimum of 1.0 ppm, ideally between 2.0-4.0 ppm, and a maximum of 4.0 ppm as per the EPA.
The required level of free chlorine in water is determined by several guidelines. According to the Association of Pool and Spa Professionals (APSP) and most other guidelines, the minimum required level of free chlorine is 1.0 parts per million (ppm). Ideally, the level of free chlorine is between 2.0 and 4.0 ppm. However, the Environmental Protection Agency (EPA) sets the maximum allowable level of free chlorine at 4.0 ppm. These levels may not be sufficient to effectively kill or control bacteria and algae or to oxidize organics.
What is the recommended level of free chlorine to prevent algae growth?
The recommended level of free chlorine to prevent algae growth is minimum 7.5% of the Cyanuric Acid (CYA) concentration. However, the EPA guidelines state that swimmers should not enter water with more than 4.0 ppm free chlorine. Therefore, the maximum CYA level in residential pool water needs to be around 50 ppm to stay within the EPA recommendation.
This recommendation is based on the understanding that the FC/CYA ratio, not the FC alone, determines the active chlorine level that prevents algae growth. For example, if a pool starts with 30 ppm of CYA, 7.5% of 30 ppm equals 2.25 ppm of FC. However, over time, the CYA level can increase to 50ppm, 80 ppm, or even 100 ppm. This would require higher FC levels: 3.75 ppm FC for 50 ppm CYA, 6.0 ppm FC for 80 ppm CYA, and 7.5 ppm FC for 100 ppm CYA. Therefore, 2.0-4.0 ppm of FC will only prevent algae when the CYA level is less than 50 ppm.
What is Combined Chlorine or Chloramines?
Combined chlorine, also known as chloramines, forms when free chlorine reacts with contaminants in the water. When free chlorine reacts with ammonia, inorganic chloramines are produced. When free chlorine reacts with organic nitrogen compounds, organic chloramines form. These chloramines cause irritation and a chlorine-like smell, particularly in indoor pools.
What is the Role of Combined Chlorine in Water Quality?
The role of combined chlorine in water quality is that it is present as a collection of contaminants that contain amine. These contaminants are inorganic and organic, originating from chemicals and people. These contaminants cause irritation in water and air, and slow down disinfection. Therefore, it is crucial to prevent and minimize its presence in pool and spa water. Combined chlorine forms when free chlorine reacts with contaminants in the water, producing either inorganic or organic chloramines, which cause evaporation and irritation to skin and mucous membranes.
What is active chlorine percentage?
Active chlorine percentage refers to the weight percentage of the active ingredient in the product. For example, a 100-pound container of trichlor would contain roughly 100 pounds of actual trichlor, as the active ingredient percentage is typically over 99%.
How to Calculate Available Chlorine Percentage?
To calculate the available chlorine percentage from test results, divide the test percent by the product’s specific gravity. This is usually listed on the product label or the SDS. For example, if the chlorine bleach test result is 14% and the specific gravity is 1.2, divide 14% by 1.2 to get 11.66% available chlorine percent.
How does chlorine interact with other chemicals in the water?
Chlorine or hypochlorous acid combine with man-made chemicals in the water. Some of these combinations lock up the chlorine, preventing it from reducing back to chloride. This removes a chloride from the water, making it unavailable for electric recycling. The only way to know the chloride level is to test the water using a chloride test strip or kit. Add more salt when the level is below the chlorine generator manufacturer’s recommended level.
What are the Properties of Chlorine?
Chlorine is 100% available and does not require a pH neutralizer. It has a pH of less than 1.0 and increases TDS by 30 ppm per 10,000 gallons, including pH neutralizer. It has an indefinite shelf life.
What are the costs of different forms of chlorine?
Gas chlorine is the cheapest and most effective sanitizer, but it is also the most dangerous and difficult to use. Liquid chlorine costs 2 times the equivalent amount of gas, calcium hypochlorite 3 times, trichlor 3 times, lithium hypochlorite 5 times, and dichlor 5 times.
What is Breakpoint Chlorination?
Breakpoint Chlorination is the process of eliminating inorganic chloramines by adding enough Free Chlorine to remove them. The formula used is ten times the Combined Chlorine minus the existing Free Chlorine.
How to achieve breakpoint chlorination?
To achieve breakpoint chlorination, add 1 gallon (3.8 liters) of anhydrous ammonia and 8 gallons (30 liters) of liquid chlorine to every 20,000 gallons (75,700 liters) of pool water. Turn off the equipment for 24 hours. After that time, add another 8 gallons of liquid chlorine, turn on the equipment, and allow swimmers back into the pool when the chlorine residual indicates normal.
What does chlorine produce when added to pool water?
When chlorine is added to water, it rapidly reacts to produce hypochlorous acid (HOCl). This acid then separates into hydrogen ion and hypochlorite ion. HOCl is the faster killing form of chlorine in water, but its effectiveness depends on the pH of the water. At a pH of 7.2, HOCl is about 65% of the FC. At a pH of 7.5, it’s about 48% of FC, and at pH 7.8, it’s about 32% of FC.
What are the Evaluation Criteria for Chlorine?
The evaluation criteria for chlorine are chlorine availability, stability, pH, convenience, and cost. Chlorine availability refers to the amount of chlorine that is available for oxidizing bacteria. Stability is about how resistant chlorine is to getting degraded by ultraviolet (UV) light. Stability also involves the actions taken to increase this resilience, ensuring the chlorine persists longer. The pH of the product is also considered in the evaluation of chlorine, because when a substantial amount is added, it changes the pH of the water. Convenience refers to how easy it is to administer the product. Lastly, the cost of the product is also a criterion.
What is an ideal Chlorine level for swimming pools?
The ideal chlorine level for swimming pools is typically recommended to be between 2.0 and 4.0 ppm (parts per million). This level is important because it controls the growth of bacteria in the pool water. Maintaining an appropriate chlorine level is essential for preventing algae and bacteria growth, ensuring the water is safe and sanitary for swimmers.
What are the signs of low Chlorine in the pool?
The signs of low chlorine in a pool are corrosive water, dissolved metals, etched plaster, eye and skin irritation, faster chlorine usage, a decrease in total alkalinity, stretching of vinyl liners, and blistering of fiberglass.
What are the consequences of swimming in a pool with low Chlorine?
Swimming in a pool with low chlorine has several consequences. Firstly, there is an increased risk of bacteria and pathogens not being effectively killed, which is a health risk to swimmers. Additionally, low chlorine levels lead to the growth of algae, resulting in cloudy and unsanitary water. Swimmers may also experience eye and skin irritation, as well as an increased risk of infections transmitted through water.
How to increase Chlorine in the pool?
To increase chlorine in the pool, the necessary chlorine for the pool size and desired chlorine level needs to be calculated. First, the desired increase in parts per million (ppm) of chlorine in the pool is to be determined. Following this, the pool’s volume in gallons is divided by 10,000. The chlorine dosage table is then referred to in order to find the quantity of chlorine needed to increase the chlorine level by 1 ppm. Lastly, the values obtained from these steps are multiplied to figure out the total quantity of chlorine required. For example, to raise the chlorine level by 3 ppm in a 18,500-gallon pool using 12.5% sodium hypochlorite, approximately 2 quarts or ½ gallon of chlorine are required.
What are the effects of low Chlorine in pool water?
The effects of low chlorine in pool water include inadequate disinfection, allowing bacteria and other microorganisms to grow. This leads to an increased risk of waterborne illnesses and infections. Low chlorine levels also result in algae growth, causing the water to become cloudy and unsafe for swimming. Additionally, low chlorine leads to poor water quality, unpleasant odors, and eye and skin irritation for swimmers.
Is low Chlorine level in the pool water dangerous?
A low chlorine level in pool water is dangerous because it is not effectively killing and controlling bacteria and algae, or adequately oxidizing organics. It is recommended to maintain a minimum free chlorine level of 1.0 ppm, an ideal range of 2.0-4.0 ppm, and a maximum of 4.0 ppm to ensure proper disinfection and prevent the growth of pathogens.
What causes low Chlorine in pool water?
The loss of chlorine in pool water is caused by several factors. One possible cause is the use of cyanuric acid, which protects the chlorine from sunlight degradation. When liquid chlorine is added to the water, it consumes the cyanuric acid in that area, leaving the remaining chlorine unprotected and susceptible to degradation. Another factor that contributes to low chlorine levels is volatilization, which is the tendency of a liquid to turn into vapor at a certain temperature. Elements such as exposure to sunlight, extent of usage, and the presence of contaminants lead to a high demand for chlorine, consequently reducing its levels in the pool water.
What are the consequences of swimming in a pool with high Chlorine?
Swimming in a pool with high chlorine has several consequences. It irritates the eyes and mucous membranes, causing discomfort and redness. It also irritates the skin, leading to dryness, itching, and rashes. Excessive levels of chlorine result in sanitizers becoming ineffective, allowing bacteria and other organisms to develop in the pool water.
How to lower pool Chlorine?
To lower the pool chlorine level, you can use the following 5 methods:
- Dilution: One way to lower the chlorine level is by diluting the pool water with fresh water. This is done by partially draining and refilling the pool.
- Natural degradation: Over time, chlorine will naturally degrade and dissipate from the pool water. By not adding any additional chlorine and allowing time to pass, the chlorine level will gradually decrease.
- Use a chlorine neutralizer: There are products available, such as chlorine neutralizers or chlorine reducers, that are added to the pool water to lower the chlorine level. These products work by chemically neutralizing the chlorine.
- Use activated carbon: Activated carbon is added to the pool’s filtration system to help remove chlorine from the water. The carbon acts as a filter and absorbs the chlorine molecules.
It’s important to note that the specific method to lower the pool chlorine level varies depending on the pool size, current chlorine level, and other factors.
How to lower Chlorine in the pool water with Dillution?
Lowering the chlorine level in pool water through dilution is achieved by mixing equal amounts of pool water and tap or bottled water, after which the test is repeated. The test result is then multiplied by 2 to obtain the accurate chlorine level. If the result of the dilution test remains high, the pool water must be diluted with further measures of bottled or tap water until the desired chlorine level is reached.”
How to lower Chlorine in the pool water with Natural degradation?
Lowering chlorine in the pool water through natural degradation is done by following these 5 steps:
- The addition of chlorine to the pool should be stopped, allowing the chlorine levels to decrease naturally over time.
- The pool’s exposure to sunlight must be increased. UV rays from the sun will breake down the chlorine in the water.
- The circulation of water in the pool must be increased to distribute the chlorine throughout the pool and promote faster degradation.
- Chlorine levels must be regularly monitored. Water testing is necessary to track the decrease in chlorine levels.
- Consideration must be given to using a chlorine neutralizer or chlorine remover product. These products can accelerate the degradation process and lower chlorine levels in the pool water.
Natural degradation of chlorine necessitates time, and the chlorine levels must be monitored until they reach the desired level.
How to lower Chlorine in the pool with chlorine reducers?
To lower chlorine levels in a pool, chlorine reducers or chlorine neutralizers must be used. These products work by chemically reacting with the chlorine in the water to reduce its concentration. The specific method of application varies depending on the chosen product. For the correct dosage and application method, it’s crucial to follow the guidelines given by the manufacturer.
What are the effects of high Chlorine in pool water?
The effects of high chlorine in pool water are skin and eye irritation, dryness or itchiness of the skin, and a strong chlorine odor. High chlorine also causes damage to swimwear and other fabrics, as well as bleaching or discoloration of hair. Additionally, high chlorine levels lead to imbalances in the pool’s pH and alkalinity levels, which result in corrosive water and damage to pool equipment.
Is high Chlorine in the pool dangerous?
Yes, high chlorine levels in a pool is dangerous. Chlorine gas is hazardous and is poisonous to humans when inhaled. Chlorine gas also irritates the eyes and mucous membranes. Additionally, high chlorine levels cause skin and hair to oxidize faster and equipment to corrode faster. For the safety of swimmers, it is important to maintain proper chlorine levels in a pool.
What are the effects of Chlorine on pool water quality?
Chlorine has several effects on pool water quality. Chlorine acts as a disinfectant, killing bacteria and other microorganisms that cause illness. Chlorine also helps to control algae growth in the pool. Additionally, chlorine reacts with organic matter in the water, such as sweat and urine, to form disinfection byproducts, such as chloramines. These byproducts cause eye and skin irritation and give the water a strong chlorine odor. It is important to maintain a balanced level of chlorine in the pool to ensure effective disinfection without causing these negative effects.
What is Chlorine in chemistry?
Chlorine is an element on the Periodic Table with the symbol Cl and atomic number 17. Chlorine is a greenish-yellow gas under standard conditions and is commonly used as a disinfectant and oxidizing agent. Chlorine is a member of the halogen group of elements and is highly reactive. Chlorine can form various compounds, including sodium hypochlorite (liquid chlorine) and trichloroisocyanuric acid (trichlor). Chlorine gas is a reference point for comparing and evaluating all other compounds that contain chlorine.
How to test swimming pool Chlorine levels in water?
To test swimming pool chlorine levels in water, chlorine test strips or a chlorine test kit are used. The test strip is dipped into the water, or a water sample is collected and the reagent from the test kit is added. After waiting for the color to develop, it is compared to the provided color chart. The resulting color indicates the level of chlorine in the water.
What is the importance of Chlorine for pool water?
Chlorine is important for pool water because it acts as a sanitizer and disinfectant, helping to kill germs and bacteria that cause illnesses. It also helps to control the growth of algae and other organic matter in the water. Chlorine is crucial for maintaining clean and safe pool water for swimmers.
How to achieve Chlorine balance in pool water?
To achieve a chlorine balance in pool water, these 8 steps must be followed:
- The desired increase in parts per million (ppm) of chlorine in the pool must be determined.
- The pool’s volume in gallons has to be divided by 10,000 to calculate the volume factor.
- The provided table hast to be referred to in order to find the amount of the specific type of chlorine that will increase the chlorine level by 1 ppm.
- The volume factor, desired increase in ppm, and the amount of chlorine needed for 1 ppm increase have to be multiplied together to calculate the total amount of chlorine needed.
- The calculated amount of chlorine has to be added to the pool water.
- Wait between 30 minutes and two hours (based on the pool volume and pool water temperature) for the chlorine to disperse and blend completely.
- The pH and alkalinity of the water hast to be tested and adjusted if necessary.
- The chlorine levels must be monitored regularly and adjustments made as needed to maintain the desired chlorine balance in the pool water.
How does sunlight affect Chlorine levels in pool water?
Sunlight significantly affects chlorine levels in pool water. When chlorine is exposed to direct sunlight, it undergoes degradation, resulting in a loss of available chlorine. The UV rays from the sun break down chlorine molecules, reducing their effectiveness in sanitizing the pool water. The rate of chlorine degradation depends on the intensity of sunlight, pH levels, presence of cyanuric acid (CYA), and the type of chlorine used.
If there is no CYA present in the pool water to protect the chlorine, 90-95% of the chlorine can be lost within 2 hours of direct sunlight exposure. This rapid degradation leads to a significant decrease in chlorine levels and a decrease in the pool’s ability to effectively sanitize the water.
In hot climates, the sun consumes up to 1.5 parts per million (ppm) of free chlorine in a day. Additionally, if swimmers apply increased amounts of sunblock, the requirement for chlorine exceeds 1.5 ppm of free chlorine per day.
To protect chlorine from sunlight degradation, it is recommended to maintain a proper CYA level in the pool water. CYA significantly slows down the UV-catalyzed degradation rate of chlorine. A cyanuric acid level of 30-50 ppm is recommended for outdoor pools.
In summary, sunlight has a detrimental effect on chlorine levels in pool water, leading to a decrease in available chlorine for sanitization. The presence of CYA improves the protection of chlorine from sunlight degradation and prolongs its effectiveness.
How does pool usage affect Chlorine levels?
The usage of a pool affects chlorine levels in several ways. If swimmers use substantial amounts of sunblock, the chlorine demand exceeds the normal rate of depletion, resulting in a decrease in chlorine levels. Additionally, the introduction of organic matter such as sweat, oils, and urine from swimmers consumes chlorine and leads to a decrease in chlorine levels. Therefore, pool usage contributes to the reduction of chlorine levels in a pool.
How often should you check Chlorine levels in your pool?
For residential use, it is recommended to check the chlorine levels in the pool weekly. However, during other periods, such as when the pool is not heavily used, it can be checked every two weeks. The frequency of checking chlorine levels in a pool varies depending on factors such as pool usage, weather conditions, and the presence of any issues or concerns. However, as a general guideline, it is recommended to check chlorine levels in a pool at least once a week. During periods of heavy usage or hot weather, it is necessary to check chlorine levels more frequently, such as every few days. To ensure effective disinfection and prevent the growth of algae and bacteria, regular monitoring and proper chlorine levels are needed.
What are the different types of Chlorine for pools?
The different types of chlorine for pools include elemental chlorine, inorganic chlorine (such as sodium hypochlorite, calcium hypochlorite, and lithium hypochlorite), and organic chlorine (such as dichlor and trichlor).
How to handle Chlorine safely when treating your pool?
To handle chlorine safely when treating the pool, it is important to follow these 9 precautions:
- Only the amount of chlorine needed for the day must be carried, and overstocking has to be avoided.
- The builder or manufacturer has to be consulted for recommendations on the type of chlorine to use, as certain types of chlorine invalidate the warranty.
- Vacuuming the stool from the pool has to be avoided; instead, the chlorine level must be increased to 10.0 ppm and the pH adjusted to between 7.2 and 7.5. The pool has to remain closed for 30 minutes.
- The desired increase in parts per million (ppm) of chlorine in the pool has to be determined and a provided table must be referred to in order to find the amount of chlorine required to achieve that increase.
- The cyanuric acid (CYA) level must be adjusted to 50 ppm using cyanuric acid or liquid instant conditioner. Granular CYA has to be used cautiously as it may take days to dissolve and potentially harm pool surfaces and equipment.
- A free chlorine level of at least 7.5% of CYA (or 5% of CYA if using 50 ppm borate) has to be maintained to avoid the need for superchlorination or the use of additional oxidizers or algaecides.
- To protect generated chlorine from sunlight degradation, a cyanuric acid level of 30-50 ppm must be maintained for outdoor pools.
- Complying with EPA guidelines is essential, and a free chlorine level of 4.0 ppm must not be exceeded to ensure the safety of swimmers.
- Chlorine has to be stored properly, stocks must be rotated, and increased vigilance regarding outside storage conditions is needed to maintain its potency.
What is the Effect of Chlorine Combining with CYA?
Chlorine combines with CYA to form new chemicals called chlorinated isocyanurates, which are not significant disinfectants or oxidizers. They are at least 150 times less effective. Most of the chlorine present in the water is chemically attached to CYA. This happens because CYA has a strong chemical attraction for chlorine, which leads them to bind together.
Are the terms hypochlorous acid and chlorine used interchangeably?
Yes, the terms are used interchangeably however Hypochlorous acid (HOCl) and chlorine are related but distinct substances. Chlorine is a chemical element, while hypochlorous acid is a compound that forms when chlorine is added to water. Hypochlorous acid is the active form of chlorine in water and is responsible for the disinfecting properties of chlorine.
Do disinfectants release chlorine in the pool?
Yes, disinfectants release chlorine in the pool.
Disinfectants like trichlor release a significant amount of chlorine when dissolved in water. For instance, a container of trichlor releases 90% of the chlorine that 100 pounds of chlorine gas would. This means that trichlor releases about 10% less chlorine than gas chlorine on a basis of equal weight. Similarly, sodium hypochlorite releases a substantial amount of chlorine, although it’s 88-90% less than gas chlorine per pound. Therefore, it is concluded that disinfectants do release chlorine in the pool.
What are the categories of chlorinating agents?
The categories of chlorinating agents are unstabilized (inorganic) and stabilized (organic). Unstabilized chlorines are sodium hypochlorite, calcium hypochlorite, lithium hypochlorite, and chlorine gas. Stabilized chlorines are trichlorostriazinetrione (also known as trichlor) and sodium dichlorostriazinetrione (also known as dichlor).
What happens to chlorine when it is used up?
When chlorine is completely used, it transforms into chloride. Chlorine changes from the disinfecting form of chlorine, hypochlorous acid (HOCl), to a harmless salt, chloride (Cl–), in the water. This is the same chloride found in table salt (sodium chloride – NaCl). Then, the chloride integrates into the total dissolved solids (TDS) present in the water and cannot be filtered out.
How does Chlorine interact with CYA?
Chlorine combines with CYA to form chlorinated isocyanurates. These new chemicals are not significant disinfectants or oxidizers. CYA has a strong affinity for chlorine, so most of the chlorine in the water is bonded to CYA. This bonding keeps the chlorine from being consumed all at once.
How are Chlorine Products Labeled?
Chlorine product labels can be misleading. They typically state the product’s purity and the percentage of available chlorine. For example, a label might read: CONCENTRATED POOL CHLORINATING GRANULES For Use in Pools, Spas, and Hot Tubs Stabilized For Extended Chlorine Life Active Ingredient: Sodium dichloroisocyanurate, anhydrous…99% Inert Ingredients…1% Total…100% (Provides 62% Available Chlorine) EPA Reg. No. 12345-15 – EPA Est. No. 12345-GA-1 NET WT. 5 Lbs. (80 oz.) (2.27 Kg) The label states that the active ingredient, sodium dichloroisocyanurate, is 99% pure. However, this doesn’t indicate the actual amount of chlorine in the product. The label also mentions 62% available chlorine, but this isn’t the total chlorine content either.
What are supplemental disinfectants?
Supplemental disinfectants are additional disinfectants used to enhance the effectiveness of primary disinfectants. They prove to be especially effective in neutralizing specific pathogens, like Giardia and Cryptosporidium, that are resistant to common pool water disinfectants like chlorine. Examples of supplemental disinfectants include ozone or ultraviolet (UV) light.
